Product Introduction
An Active Pharmaceutical Ingredient (API) is the primary component in a drug that produces the intended therapeutic effect. In any pharmaceutical formulation, the API is the biologically active substance responsible for treating, diagnosing, or preventing disease. APIs are manufactured through various processes, including chemical synthesis, fermentation, and biotechnological methods. High-purity APIs are essential in ensuring the safety, efficacy, and quality of medications.
-
Calcium Dobesilate丨CAS 20123-80-2Catalog No.: SS144465. CAS No.: 20123-80-2. Grade: EP11.0. Product Name: Calcium dobesilate. Molecular Formula: (C6H5O5S)2.Ca; C12H10Ca2O10S2. Molecular Weight: 418.41.read more
-
Lisinopril丨CAS 83915-83-7Catalog No.: SS143223 . CAS No.: 83915-83-7 . Grade: EP10.0. Product Name: Lisinopril. Molecular Formula: C21H31O5N3.2(H2O). Molecular Weight: 441.52 . Synonym(s): (S)-1-(N2-(1-Carboxy-3-phenylpropyl)-L-lysyl)-L-proline dihydrate.read more
-
Baricitinib丨CAS 1187594-09-7Catalog No.: SS093544. CAS No.: 1187594-09-7. Assay: 98%-102% . Product Name: Baricitinib. Molecular Formula: C16H17N7O2S. Molecular Weight: 371.4.read more
-
Cefadroxil Monohydrate丨CAS 66592-87-8Catalog No.: SS142292. CAS No.: 66592-87-8 . Grade: BP2025. Product Name: Cefadroxil Monohydrate. Molecular Formula: C16H17N3O5S.H2O. Molecular Weight: 381.41.read more
-
Minocycline Hydrochloride丨CAS 13614-98-7Catalog No.: SS142211. CAS No.: 13614-98-7. Grade: EP grade. Product Name: Minocycline hydrochloride. Molecular Formula: C23H27N3O7.HCl. Molecular Weight: 493.94.read more
-
Carbidopa Monohydrate丨CAS 38821-49-7Catalog No.: SS085937. CAS No.: 38821-49-7. Grade: USP43. Product Name: Carbidopa Monohydrate. Molecular Formula: C10H16N2O5. Molecular Weight: 244.25. Synonym(s): (S)-3-(3,4-Dihydroxyphenyl)-2-hydrazino-2-methylpropionic Acid.read more
-
Heparin Sodium丨CAS 9041-08-1Catalog No.: SS111990. CAS No.: 9041-08-1. Grade: EP 11.0. Product Name: Heparin sodium . Certificate: ISO9001.read more
-
Perindopril Arginine丨CAS 612548-45-5Catalog No.: SS141637. CAS No.: 612548-45-5. Assay (HPLC): 98.0%~102.0% (On the anhydrous and solvent-free basis). Product Name: Perindopril arginine. Molecular Formula: C25H46N6O7. Molecular Weight: 542.7. Synonym(s): Perindoprilread more
-
Kanamycin Sulfate丨CAS 25389-94-0Catalog No.: SS141626. CAS No.: 25389-94-0 . Grade/Potency (Calculated by dry product): CP2000/BP2025/765u/mg min. Product Name: Kanamycin sulfate. Molecular Formula: C18H36N4O11.H2SO4; C18H38N4O15S. Molecular Weight: 582.57. Synonym(s):read more
-
Dypyridamole丨CAS 58-32-2Catalog No.: SS141247. CAS No.: 58-32-2 . Grade: EP11.0. Product Name: Dypyridamole. Certificate: ISO14001. Molecular Formula: C24H40N8O4. Molecular Weight: 504.63 . Synonym(s):read more
-
Trilostane丨CAS 13647-35-3Catalog No.: SS110071 . CAS No.: 13647-35-3. Assay: 99.0% min. Product Name: Trilostane . Molecular Formula: C20H27NO3. Molecular Weight: 329.43. Synonym(s): (4a,5a,17b)-3,17-Dihydroxy-4,5-epoxyandrost-2-ene-2-carbonitrile .read more
-
Piroxicam丨CAS 36322-90-4Catalog No.: SS035883. CAS No.: 36322-90-4. Grade: USP42. Product Name: Piroxicam . Molecular Formula: C15H13N3O4S . Molecular Weight: 331.34. Synonym(s): 4-Hydroxy-2-methyl-N-2-pyridinyl-2H-1,2-benzothiazine-3-carboxamide 1,1-dioxide.read more
Product advantages and features
Therapeutic Efficacy
APIs are the core components that provide the drug’s intended medicinal effects.
01
Precision and Consistency
Produced under strict regulatory standards, ensuring consistent potency and purity across batches.
02
Wide Application in Drug Formulation
APIs are used in various dosage forms like tablets, capsules, injectables, and topical formulations.
03
Customization
APIs can be modified and optimized for different routes of administration, enhancing drug performance.
04
Global Compliance
APIs are manufactured according to GMP, FDA, and other international standards, ensuring they meet stringent quality and safety requirements.
05
Product type
Manufactured through chemical synthesis, commonly used in most conventional drugs.
Produced from living organisms using biotechnological processes; commonly used in vaccines, monoclonal antibodies, and other biologics.
Derived from natural sources like plants, minerals, or marine organisms, these are used in herbal or traditional medicines.
Used in specialized treatments like oncology, where very small doses provide significant therapeutic effects.
Chemically identical to branded APIs but produced for off-patent drugs, making them cost-effective alternatives.
Application of the product
Pharmaceutical Manufacturing: APIs are the central ingredients in creating various medications used for treating numerous health conditions.
Research and Development: APIs are critical in drug discovery, testing, and clinical trials for developing new therapeutics.
Personalized Medicine: Tailored APIs are used in customized treatments that cater to individual patient needs.
Generic Drug Production: Widely used in the production of generic medications after patents expire, making treatments more accessible.
Biopharmaceuticals: APIs are essential in advanced biologics, including gene therapy and advanced cancer treatments.
Material of the product
Organic Compounds: Most APIs are based on organic molecules, synthesized to achieve specific medicinal properties.
Biological Materials: APIs in biopharmaceuticals are derived from proteins, enzymes, or other biomolecules produced via recombinant DNA technology.
Mineral Sources: Some APIs, like those used in supplements, are derived from minerals and are purified for medical use.
Plant Extracts: Natural APIs derived from plants are often used in traditional and alternative medicine.
EPeptides and Oligonucleotides: Specialized materials used in targeted therapies and advanced drug formulations.
Production Process Or Procedure
API manufacturing begins with the sourcing of raw materials, which are then subjected to chemical synthesis or biotechnological processes. The synthesis involves complex reactions, purification steps, and refining to achieve the desired purity and potency. After production, APIs undergo rigorous quality control, including tests for impurities, stability, and potency. The final product is then packaged, labeled, and prepared for distribution to pharmaceutical companies for formulation.
Components of the product
Active Substance
The main therapeutic component that delivers the medicinal effect.
Impurities and By-products
Controlled and minimized during the production process to meet safety standards.
Stabilizers
Added to ensure the API remains stable during manufacturing and storage.
Solvents
Used during synthesis, carefully removed during purification.
Isomeric Forms
In some cases, APIs may consist of specific isomers or enantiomers that exhibit the desired biological activity.
Product maintenance and precautions
Store APIs in controlled environments with specific temperature and humidity levels to prevent degradation.
Use protective equipment when handling APIs, especially HPAPIs, which require containment strategies.
Regularly test batches for potency, purity, and stability to ensure consistent quality.
Follow all local and international guidelines for API handling, storage, and transportation.
Track expiration dates and perform stability testing to guarantee the API remains effective over time.
Company advantages
Our Focus on Quality & Customer Experience
We dedicate to delivering high-quality products and exceptional customer service beyond your expectation.
Comprehensive Product Range
Extensive catalog of over 27,000 chemicals for diverse industries, offering both standard compounds and specialized solutions.
Strong Industry Expertise
Our experienced diverse team provides expert guidance, staying current with industry trends to deliver optimal chemical solutions tailored to your needs.
Competitive Pricing & Reliability
Competitive pricing and reliable deliveries ensure cost-effective, efficient supply chains without compromising quality.
How to collaborate with us
Partnering with LEAPChem gives you access to the expertise of a leading company with 18 years of experience in the chemical industry. We offer over 27,000 chemical substances tailored to meet diverse needs. All our products adhere to industry standards and are accompanied by the necessary certifications.
We collaborate with a wide range of partners, including:
• Research Laboratories
• Pharmaceutical Companies
• Cosmetics Manufacturers
• Electronics Manufacturers
• Chemical Manufacturers
• Universities and Colleges Worldwide
• And more.
We specialize in helping our clients find the right chemical solutions tailored to their specific requirements. Additionally, we provide custom synthesis services to meet the unique needs of our partners. Whether you need bulk quantities for industrial processes or small batches for specialized applications, we’ve got you covered.
If you are looking for a reliable chemical supplier, we would be delighted to hear from you. Please send your inquiry or project details to sales@leapchem.com. Our professional team is eager to collaborate with you and help achieve your goals.
FAQ
Q: What is an API in pharmaceuticals?
Q: How are APIs manufactured?
Q: What is the difference between an API and a drug?
Q: What is the importance of purity in APIs?
Q: Are there different types of APIs?
Q: How are APIs tested for quality?
Q: What are HPAPIs, and where are they used?
Q: How are APIs stored?
Q: What are the global regulations governing API production?
Q: Can APIs be customized for specific drug formulations?
Q: What role do APIs play in generic drug production?
Q: What are biological APIs?
Q: What are the challenges in API manufacturing?
Q: How are APIs used in personalized medicine?
Q: What testing methods ensure API quality?
Q: Why is the manufacturing environment critical for APIs?
Q: How do biopharmaceuticals utilize APIs?
Q: What is the significance of generic APIs?
Q: What safety measures should be taken when handling APIs?
Q: How do APIs contribute to drug research and development?
Hangzhou Leap Chem Co., Ltd. is well-known as one of the leading api manufacturers and suppliers in China. We warmly welcome you to wholesale cheap api from our factory. Contact us for custom service.
Metronidazole, 443-48-1, Metformin hydrochloride-
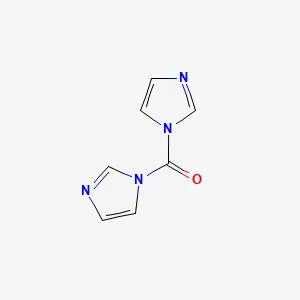
1,1'-Carbonyldiimidazole丨CAS 530-62-1Catalog No.: SS080884read more
CAS No.: 530-62-1
Assay(T): 98%min
Product Name:... -
Palladium丨CAS 7440-05-3Catalog No.: SS067788read more
CAS No.: 7440-05-3
Assay(Pd): 10% Pd/C on dry basis.
Product Name:... -
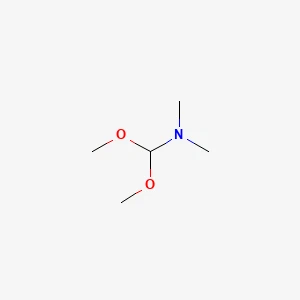
N,N-Dimethylformamide Dimethyl Acetal丨CAS 4637-24-5Catalog No.: SS036295read more
CAS No.: 4637-24-5
Assay: 98%min
Product Name: N,N-Dimethylformamide... -
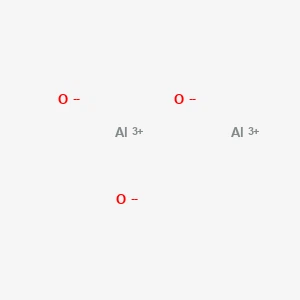
Aluminum Oxide丨CAS 1344-28-1Catalog No.: SS119815read more
CAS No.: 1344-28-1
Purity: 99.99% min; Average particle size: 50nm... -
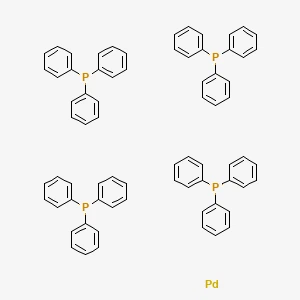
Tetrakis(triphenylphosphine)palladium丨CAS 14221-01-3Catalog No.: SS118525read more
CAS No.: 14221-01-3
Purity: 99%min Pd: 9.2%min
Product Name:... -
Alpha-Arbutin丨CAS 84380-01-8Catalog No.: SS087082read more
CAS No.: 84380-01-8
Purity: 99.5%min.
Product Name: alpha-Arbutin... -
N-Iodosuccinimide丨CAS 516-12-1Catalog No.: SS018455read more
CAS No.: 516-12-1
Purity: 98.0%min
Product Name: N-Iodosuccinimide... -
Octadecanethiol丨CAS 2885-00-9Catalog No.: SS085366read more
CAS No.: 2885-00-9
Assay(GC): 98.00%min
Product Name: Octadecanethiol...