Product Introduction
Pharmaceutical excipients are inactive substances used in drug formulations to serve as carriers, fillers, or stabilizers, enhancing the performance and delivery of the active pharmaceutical ingredient (API). Though excipients do not provide therapeutic effects, they play critical roles in ensuring the safety, effectiveness, and manufacturability of the drug product. From enhancing solubility to controlling drug release, excipients are indispensable in the production of tablets, capsules, injectables, and other dosage forms.
-
Glyceryl Dibehenate丨CAS 99880-64-5Catalog No.: SS143666. CAS No.: 99880-64-5. Grade: EP10.0. Product Name: Glyceryl Dibehenate. Molecular Formula: C12H14O3. Molecular Weight: 206.24. Synonym(s): Docosanoic acid, diester with 1,2,3-propanetriol.read more
-
Sesame Oil丨CAS 8008-74-0Catalog No.: SS143687. CAS No.: 8008-74-0 . Grade: USP NF2021/EP11.0. Product Name: Sesame Oil. Certificate: DMF.read more
-
Tert-Butanol丨CAS 75-65-0Catalog No.: SS140682. CAS No.: 75-65-0 . Grade: Pharma grade. Product Name: tert-Butanol. Molecular Formula: C4H10O. Molecular Weight: 74.12 . Synonym(s): tert-Butyl alcohol; 1,1-Dimethylethanol; 2-Methyl-2-propanol.read more
-
Manganese Chloride Tetrahydrate丨CAS 13446-34-9Catalog No.: SS138919. CAS No.: 13446-34-9. Grade/Assay (MnCl2, on dry basis): Pharma Grade/98.0%~101.0%. Product Name: Manganese chloride tetrahydrate. Molecular Formula: MnCl2.4(H2O). Molecular Weight: 197.91. Synonym(s): Manganese(II)read more
-
Zinc Oxide丨CAS 1314-13-2Catalog No.: SS132995. CAS No.: 1314-13-2. Grade/Assay (ZnO, on dry basis): CP/99.0% min . Product Name: Zinc oxide. Certificate: WC. Molecular Formula: ZnO. Molecular Weight: 81.39.read more
-
Poloxamer 188丨CAS 9003-11-6Catalog No.: SS137660 . CAS No.: 9003-11-6. Grade: EP grade; injection grade . Product Name: Poloxamer 188. Certificate: DMF. Molecular Formula: (C3H6O · C2H4O)x. Synonym(s): Poly(ethylene glycol)-block-poly(propyleneread more
-
Poloxamer 182丨CAS 9003-11-6Catalog No.: SS136946. CAS No.: 9003-11-6. Grade/ Average molecular weight: Pharmaceutical Grade/1700~3200. Product Name: Poloxamer 182 . Certificate: ISO9001; ISO14001 . Molecular Formula: C7H16O4 . Molecular Weight: 164.20.read more
-
Sodium Carboxyl Methylstarch丨CAS 9063-38-1Catalog No.: SS135490 . CAS No.: 9063-38-1. Grade: EP10.0 potato base. Product Name: Sodium carboxyl methylstarch. Molecular Formula: C11H14FNO3S. Molecular Weight: 259.30. Synonym(s): Sodium Starch Glycolate .read more
-
Chitosan, 2-hydroxypropyl Ether丨CAS 84069-44-3Catalog No.: SS129620. CAS No.: 84069-44-3. Assay: 98% min . Product Name: Chitosan, 2-hydroxypropyl ether. Molecular Formula: C51H101N3O24. Molecular Weight: 1140.35334. Synonym(s): Chitofilmer.read more
-
Hydrogenated Palm Glycerides丨CAS 91744-66-0Catalog No.: SS130261. CAS No.: 91744-66-0. Saponification value: 195~210 . Product Name: Hydrogenated palm glycerides.read more
-
Calcium Stearate丨CAS 1592-23-0Catalog No.: SS084965 . CAS No.: 1592-23-0 . Grade: USP35-NF30. Product Name: Calcium stearate. Certificate: ISO9001; Halal; Kosher. Molecular Formula: CH3(CH2)16COO2Ca. Molecular Weight: 607.03. Synonym(s): Octadecanoic acid calcium salt;read more
-
Calcium Silicate丨CAS 1344-95-2Catalog No.: SS131685. CAS No.: 1344-95-2. Grade: CP 2020. Product Name: Calcium silicate. Molecular Formula: CaH6O8Si2. Molecular Weight: 230.29. Synonym(s): Calcarea silicata.read more
Product advantages and features
Improved Drug Stability
Excipients help maintain the stability of the API during manufacturing, storage, and administration.
01
Enhanced Drug Delivery
They enhance the bioavailability of the API, ensuring consistent and efficient drug absorption.
02
Controlled Release Mechanisms
Excipients allow for sustained or delayed release of drugs, optimizing therapeutic outcomes.
03
Manufacturability and Processability
They facilitate the efficient manufacturing of various dosage forms by improving flowability, compressibility, and product uniformity.
04
Patient-Centric Solutions
Excipients can improve the taste, texture, and appearance of drugs, making them more acceptable and easier to administer.
05
Product type
These excipients hold the components of a tablet together, ensuring structural integrity and consistent dosing.
Added to promote the breakdown of tablets into smaller fragments in the gastrointestinal tract, ensuring rapid drug release.
Used to bulk up formulations with small quantities of API, making tablets or capsules a practical size.
Excipients like magnesium stearate are added to reduce friction during the tablet manufacturing process, preventing sticking and ensuring smooth production.
Used in applications requiring see-through displays, like heads-up displays in vehicles and smart windows.Applied to protect the tablet, mask unpleasant tastes, and sometimes control the drug’s release profile.
Application of the product
Tablet and Capsule Formulation: Excipients are essential for producing solid oral dosage forms, ensuring proper tablet structure, stability, and drug release.
Injectable Formulations: In injectables, excipients act as solvents, stabilizers, or pH adjusters, ensuring the API remains effective and safe for administration.
Topical and Transdermal Products: Excipients are used to enhance absorption through the skin and improve the texture and feel of creams, gels, and ointments.
Controlled Release Systems: Specialized excipients enable sustained or delayed drug release, allowing for fewer doses and improved patient adherence.
Nutraceuticals and Dietary Supplements: Excipients are also widely used in supplements to ensure consistent quality, stability, and ease of consumption.
Material of the product
Cellulose Derivatives: Widely used as binders, fillers, and disintegrants in tablet formulations, providing flexibility in drug design.
Polyethylene Glycol (PEG): A versatile excipient used in various dosage forms for solubilizing and stabilizing APIs.
Starch and Starch Derivatives: Commonly used as fillers and disintegrants, contributing to the overall performance and stability of solid oral dosage forms.
Silica-Based Compounds: These are often used as glidants to improve powder flow during the manufacturing process.
Sugars and Polyols: Used as sweeteners or bulking agents, enhancing the taste and mouthfeel of oral formulations.
Production Process Or Procedure
Pharmaceutical excipients are produced through both natural and synthetic processes. Natural excipients like starch are extracted and purified, while synthetic excipients like PEG are manufactured through chemical synthesis. Once produced, excipients undergo rigorous quality control tests, including assessments for purity, consistency, and stability. The selection and combination of excipients are tailored based on the specific needs of the drug formulation, ensuring optimal performance and safety in the final product.
Components of the product
Carrier Agents
The primary materials that support or carry the API in the formulation, ensuring even distribution.
Stabilizers
These prevent the degradation of the API due to environmental factors like light, temperature, or moisture.
Flow Aids
Excipients that improve the flow properties of powders during manufacturing, reducing clumping and ensuring uniformity.
Taste Masking Agents
Ingredients added to mask the bitter or unpleasant taste of APIs, improving patient compliance.
Preservatives
Excipients that prevent microbial growth, especially in liquid formulations, extending the shelf life of the product.
Product maintenance and precautions
Excipients should be stored in controlled environments, protecting them from moisture, heat, and contamination.
Excipients must be handled according to GMP guidelines to avoid cross-contamination and degradation.
Regular quality control tests should be conducted to ensure excipients maintain their intended properties throughout storage and use.
Follow regulatory guidelines (e.g., FDA, ICH) regarding excipient use in pharmaceuticals to ensure safety and quality.
Excipients must be tested for compatibility with APIs and other formulation components to avoid unwanted interactions or reduced efficacy.
Company advantages
Our Focus on Quality & Customer Experience
We dedicate to delivering high-quality products and exceptional customer service beyond your expectation.
Comprehensive Product Range
Extensive catalog of over 27,000 chemicals for diverse industries, offering both standard compounds and specialized solutions.
Strong Industry Expertise
Our experienced diverse team provides expert guidance, staying current with industry trends to deliver optimal chemical solutions tailored to your needs.
Competitive Pricing & Reliability
Competitive pricing and reliable deliveries ensure cost-effective, efficient supply chains without compromising quality.
How to collaborate with us
Partnering with LEAPChem gives you access to the expertise of a leading company with 18 years of experience in the chemical industry. We offer over 27,000 chemical substances tailored to meet diverse needs. All our products adhere to industry standards and are accompanied by the necessary certifications.
We collaborate with a wide range of partners, including:
• Research Laboratories
• Pharmaceutical Companies
• Cosmetics Manufacturers
• Electronics Manufacturers
• Chemical Manufacturers
• Universities and Colleges Worldwide
• And more.
We specialize in helping our clients find the right chemical solutions tailored to their specific requirements. Additionally, we provide custom synthesis services to meet the unique needs of our partners. Whether you need bulk quantities for industrial processes or small batches for specialized applications, we’ve got you covered.
If you are looking for a reliable chemical supplier, we would be delighted to hear from you. Please send your inquiry or project details to sales@leapchem.com. Our professional team is eager to collaborate with you and help achieve your goals.
FAQ
Q: What are pharmaceutical excipients?
Q: Why are excipients necessary in drug formulations?
Q: What types of excipients are commonly used in tablets?
Q: How do excipients enhance drug solubility?
Q: What is the difference between an excipient and an API?
Q: Are pharmaceutical excipients safe?
Q: Can excipients affect drug release rates?
Q: How are excipients selected for a drug formulation?
Q: What role do excipients play in injectable drugs?
Q: What are some common natural excipients?
Q: How are pharmaceutical excipients regulated?
Q: Can excipients cause allergies or adverse reactions?
Q: What is the role of preservatives in pharmaceutical formulations?
Q: How do excipients improve patient compliance?
Q: Are there specialized excipients for controlled release formulations?
Q: What is the importance of batch-to-batch consistency in excipients?
Q: How do excipients impact the shelf life of a drug?
Q: Are excipients used in dietary supplements as well?
Q: What are filler excipients, and why are they used?
Q: Can excipients be used to enhance the aesthetics of pharmaceutical products?
Hangzhou Leap Chem Co., Ltd. is well-known as one of the leading pharmaceutical excipients manufacturers and suppliers in China. We warmly welcome you to wholesale cheap pharmaceutical excipients from our factory. Contact us for custom service.
CAS 12650 88 3, pharmaceutical excipients, 12650-88-3-
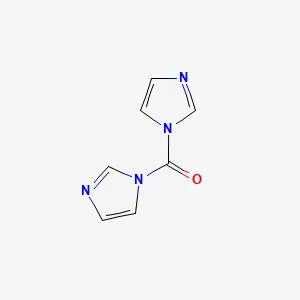
1,1'-Carbonyldiimidazole丨CAS 530-62-1Catalog No.: SS080884read more
CAS No.: 530-62-1
Assay(T): 98%min
Product Name:... -
Palladium丨CAS 7440-05-3Catalog No.: SS067788read more
CAS No.: 7440-05-3
Assay(Pd): 10% Pd/C on dry basis.
Product Name:... -
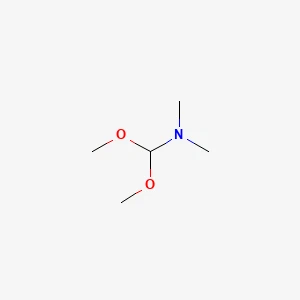
N,N-Dimethylformamide Dimethyl Acetal丨CAS 4637-24-5Catalog No.: SS036295read more
CAS No.: 4637-24-5
Assay: 98%min
Product Name: N,N-Dimethylformamide... -
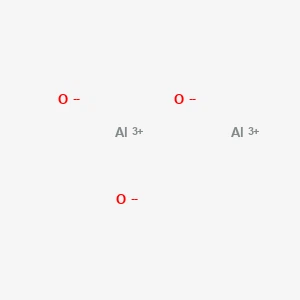
Aluminum Oxide丨CAS 1344-28-1Catalog No.: SS119815read more
CAS No.: 1344-28-1
Purity: 99.99% min; Average particle size: 50nm... -
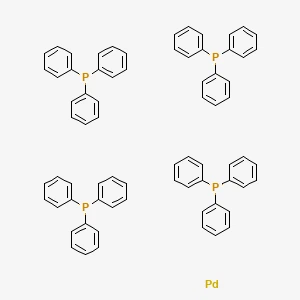
Tetrakis(triphenylphosphine)palladium丨CAS 14221-01-3Catalog No.: SS118525read more
CAS No.: 14221-01-3
Purity: 99%min Pd: 9.2%min
Product Name:... -
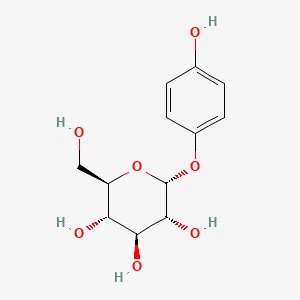
Alpha-Arbutin丨CAS 84380-01-8Catalog No.: SS087082read more
CAS No.: 84380-01-8
Purity: 99.5%min.
Product Name: alpha-Arbutin... -
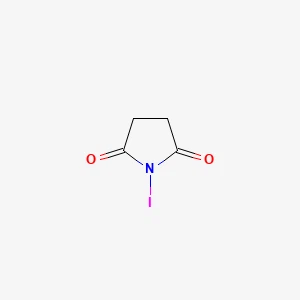
N-Iodosuccinimide丨CAS 516-12-1Catalog No.: SS018455read more
CAS No.: 516-12-1
Purity: 98.0%min
Product Name: N-Iodosuccinimide... -
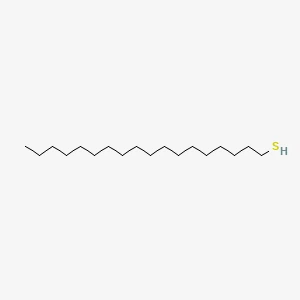
Octadecanethiol丨CAS 2885-00-9Catalog No.: SS085366read more
CAS No.: 2885-00-9
Assay(GC): 98.00%min
Product Name: Octadecanethiol...