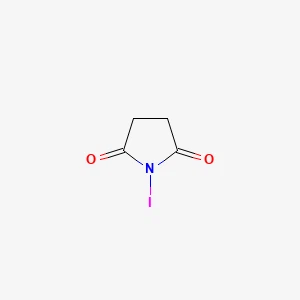
Hangzhou Leap Chem Co., Ltd. is one of the most professional manufacturers and suppliers of n-iodosuccinimide丨cas 516-12-1 in China. Welcome to wholesale custom made chemical products at competitive price from our factory. For more cheap products, contact us now.
Specifications of N-Iodosuccinimide丨516-12-1
|
Property |
Specification |
|
Appearance |
White or light yellow crystalline powder |
|
Purity |
98.0% min |
|
Active iodine |
55~58% |
|
Loss on drying |
0.5% max |
|
MP (Melting point) |
192 to 203°C |
Transport Information of N-Iodosuccinimide丨516-12-1
|
Parameter |
Specification |
|
UN Number |
|
|
Class |
|
|
Packing Group |
|
|
H.S. Code |
2933990099306 |
|
Stability & Reactivity |
Stable |
|
Storage |
2~8 deg.C |
|
Condition to Avoid |
|
|
Package |
Introduction
N-Iodosuccinimide丨516-12-1, is a highly useful reagent in organic chemistry, particularly for selective iodination and oxidation reactions. It is commonly used in the synthesis of iodinated compounds and other halogenated organic molecules. N-Iodosuccinimide is a solid compound with the chemical formula C₄H₆INO₂, and it consists of a succinimide molecule with an iodine atom attached to it.
Below are the key applications and benefits of N-Iodosuccinimide丨516-12-1:
1. Iodination Reactions
● Selective Iodination: N-Iodosuccinimide is most commonly used as an iodinating agent. It provides a controlled source of iodine, which can be used to selectively introduce iodine atoms into organic molecules. Iodination reactions are often more selective and mild compared to other halogenation methods, making NIS particularly useful in situations where specific iodination is required without affecting other functional groups.
● Iodination of Aromatic Compounds: NIS is frequently used in aryl iodination reactions, where it reacts with aromatic compounds to replace a hydrogen atom with an iodine atom. This is particularly useful in the synthesis of iodinated aromatics, which have applications in pharmaceuticals, agrochemicals, and materials science.
● Iodination of Aliphatic Compounds: NIS can also iodinate aliphatic compounds, although its primary use is in iodination of aromatic systems. The reactivity and selective nature of NIS make it valuable for introducing iodine atoms into complex organic molecules with precision.
2. Oxidation Reactions
● Oxidative Reactions: N-Iodosuccinimide is used in oxidation reactions to convert certain functional groups into their oxidized forms. For example, it can be used to oxidize alcohols to aldehydes or aldehydes to acids. The reagent is often favored over other oxidants because it is relatively mild and selective.
● Oxidation of Sulfur Compounds: It is also effective for the oxidation of sulfur-containing compounds, such as thiols or sulfides, to sulfoxides or sulfones. This selective oxidation can be useful in the synthesis of bioactive molecules or functional materials.
● Hydroxylation of Alkenes: NIS has been used in the hydroxylation of alkenes, where it adds a hydroxyl group to a carbon-carbon double bond. This transformation is useful in the creation of alcohol-containing compounds.
3. Radical Reactions
● Radical Iodination: N-Iodosuccinimide丨516-12-1 is often employed in radical-mediated iodination reactions. Under the right conditions, it can generate iodine radicals (I•) that are highly reactive and can participate in chain reactions, adding iodine atoms to a variety of substrates. This makes NIS valuable in reactions where radical mechanisms are involved.
● Halogenation in the Presence of Light: In the presence of ultraviolet (UV) light or heat, NIS can undergo homolytic cleavage, generating iodine radicals that can initiate halogenation reactions. This feature is particularly useful in the selective halogenation of organic molecules under mild conditions.
4. Synthesis of Iodinated Organic Compounds
● Iodinated Pharmaceuticals: N-Iodosuccinimide is used to iodinate pharmaceutical intermediates and active pharmaceutical ingredients (APIs). Iodine substitution can significantly alter the properties of a molecule, including its biological activity, metabolic stability, and bioavailability. Therefore, NIS is valuable in drug development, especially when fluorine or iodine incorporation is required for better efficacy or selectivity.
● Agrochemical Synthesis: Iodinated organic compounds are important in the agrochemical industry, where iodine atoms are often incorporated into herbicides, fungicides, and insecticides. N-Iodosuccinimide serves as a key reagent in the synthesis of such iodinated agrochemicals.
5. Functional Group Transformations
● Halogenation of Ketones and Aldehydes: N-Iodosuccinimide丨516-12-1 is useful for the halogenation of carbonyl compounds, such as ketones and aldehydes. The iodine atom can be selectively introduced at the α-position relative to the carbonyl group, which is important for subsequent functional group transformations.
● Modification of Amines: N-Iodosuccinimide can react with amines to form iodoamine derivatives. This is useful in the preparation of iodo-substituted amines, which can be further functionalized or used as building blocks in the synthesis of complex molecules.
6. Synthesis of Organoiodine Compounds
● Preparation of Organoiodine Intermediates: N-Iodosuccinimide is used in the synthesis of organoiodine intermediates that are essential in the production of various organic molecules. These intermediates are crucial in the formation of carbon-iodine bonds, which can be further modified in numerous organic transformations.
● Cross-Coupling Reactions: Iodine-substituted organic molecules can be used in cross-coupling reactions such as Suzuki, Heck, or Sonogashira reactions, where the carbon-iodine bond is used to couple organic fragments. NIS provides a selective and efficient method for introducing iodine into organic molecules, making them suitable for these coupling reactions.
7. Benefits of N-Iodosuccinimide丨516-12-1
● Mild and Selective Iodination: N-Iodosuccinimide is favored in organic synthesis because it provides a mild and selective iodination method. The iodine atom is introduced without requiring harsh conditions, making it a safer alternative to other iodination reagents such as iodine or iodine monochloride (ICl).
● Controlled Release of Iodine: NIS offers controlled release of iodine, which makes it suitable for reactions where precise amounts of iodine are needed, as in the synthesis of iodinated bioactive molecules or in highly selective iodination reactions.
● Versatility: NIS can be used in a wide range of reactions, including oxidation, iodination, and functional group transformations. Its versatility makes it a valuable reagent in both laboratory and industrial settings.
● Non-Toxic Byproducts: Unlike some other halogenating reagents, N-Iodosuccinimide decomposes into non-toxic byproducts such as succinimide, which is not harmful and can be easily removed from reaction mixtures. This makes NIS a relatively safer alternative for iodination in chemical processes.
8. Safety Considerations
● Handling: N-Iodosuccinimide丨516-12-1 is generally safe to handle under controlled conditions, but it should be handled with care due to its reactivity. It should be stored in a cool, dry place, away from moisture or incompatible substances.
● Irritant: Like many halogenated reagents, NIS can be an irritant to the skin, eyes, and respiratory system. Proper personal protective equipment (PPE), such as gloves, goggles, and a lab coat, should be worn when handling NIS. It should also be used in well-ventilated areas or under a fume hood to avoid inhalation of any vapors.
● Reaction with Water: N-Iodosuccinimide should not be exposed to moisture or water, as it may hydrolyze, potentially releasing toxic iodine vapors. It is important to ensure that the reagent is kept dry during use and storage.
Conclusion
N-Iodosuccinimide丨516-12-1 is a highly useful reagent in organic synthesis, particularly for iodination reactions, oxidation of functional groups, and the preparation of iodinated intermediates in pharmaceutical, agrochemical, and materials science. Its mild, selective, and controlled release of iodine makes it an essential tool in many synthetic chemistry applications. Whether used for the synthesis of iodinated compounds, radical reactions, or functional group transformations, NIS offers significant advantages in efficiency and precision. With proper safety precautions, N-Iodosuccinimide provides a reliable and versatile method for halogenation and oxidation reactions in both academic and industrial settings.