Product Introduction
Crown ethers are a group of synthetic cyclic chemical compounds known for their ability to form stable complexes with various metal ions. These macrocyclic compounds are named "crown" due to their circular structure, which resembles a crown when complexed with ions. Crown ethers are used extensively in chemical synthesis, ion transport, and separation processes due to their unique ability to selectively bind cations, particularly alkali and alkaline earth metals. This selectivity makes them highly valuable in organic chemistry, pharmaceutical research, and industrial applications where ion recognition and transport are crucial.
-
Benzo-18-crown-6丨CAS 14098-24-9Catalog No.: SS113713. CAS No.: 14098-24-9 . Purity(GC): 98% min. Product Name: Benzo-18-crown-6 . Molecular Formula: C16H24O6. Molecular Weight: 312.36.read more
-
12-Crown-4丨CAS 294-93-9Catalog No.: SS132255. CAS No.: 294-93-9. Purity: 99% min . Product Name: 12-Crown-4. Molecular Formula: C8H16O4. Molecular Weight: 176.21. Synonym(s): 1,4,7,10-Tetraoxacyclododecane.read more
-
18-Crown-6丨CAS 17455-13-9Catalog No.: SS057205CAS No.: 17455-13-9Assay: 99% min Product Name: 18-Crown-6Molecular Formula: C12H24O6Molecular Weight: 264.32Synonym(s): 1,4,7,10,13,16-Hexaoxacyclooctadecane; Hexaoxacyclo-octadecane; 18-Crown 6-etherread more
-
Benzo-15-crown-5丨CAS 14098-44-3Catalog No.: SS113528. CAS No.: 14098-44-3. Purity(GC): 99% min. Purity(HPLC): 99% min . Product Name: Benzo-15-crown-5. Molecular Formula: C14H20O5. Molecular Weight: 268.31. Synonym(s): 1,4,7,10,13-Benzopentaoxacyclopentadecin,read more
-
Dibenzo-18-crown-6丨CAS 14187-32-7Catalog No.: SS110025. CAS No.: 14187-32-7. Purity(HPLC): 98%min. Product Name: Dibenzo-18-crown-6. Molecular Formula: C20H24O6. Molecular Weight: 360.41. Synonym(s): Dibenzo-18-crown-6-ether.read more
Product Advantages And Features
Selective Cation Binding
Crown ethers can selectively bind specific metal ions, making them useful in separation and purification processes, particularly in industries like pharmaceuticals and water treatment.
Facilitates Chemical Reactions
By complexing with metal cations, crown ethers enhance the solubility of salts in organic solvents, promoting a wide variety of reactions, especially in organic synthesis.
Applications in Green Chemistry
Crown ethers are utilized in environmentally friendly chemical processes as they can reduce the need for harmful solvents and reagents by simplifying reaction mechanisms.
High Thermal Stability
Crown ethers possess high thermal stability, allowing them to be used in high-temperature chemical processes without degradation.
Wide Range of Sizes and Types
Different sizes and structures of crown ethers allow for tailored use in binding different metal ions, providing flexibility for a range of chemical applications.
Product Type
This small crown ether binds with smaller ions, such as lithium (Li+), and is typically used in specialized organic reactions where smaller cations are involved.
15-Crown-5 is known for its ability to selectively bind sodium ions (Na+), making it useful in ion separation and organic synthesis. It is also commonly employed in the stabilization of reactive intermediates in organic chemistry.
One of the most common crown ethers, 18-Crown-6 is especially effective at binding potassium ions (K+), widely used in phase transfer catalysis and facilitating nucleophilic reactions in nonpolar solvents.
These are crown ethers with benzene rings integrated into their structure. Dibenzo crown ethers are often more selective and are used in complexation reactions involving specific ions, providing enhanced binding properties.
Azacrown ethers contain nitrogen atoms in their ring structure, which allows them to bind not only metal cations but also protons, making them suitable for both cation and proton transport in chemical systems.
Application Of The Product
Phase Transfer Catalysis: Crown ethers facilitate the transfer of ionic species from aqueous to organic phases, enabling reactions between compounds that would otherwise be immiscible. This application is key in organic synthesis, especially in the production of complex molecules.
Ion Transport and Separation: Crown ethers are used to selectively transport metal ions across membranes, which is important in processes like metal ion extraction and purification, as well as in battery technology and ion sensors.
Synthesis of Pharmaceuticals: In pharmaceutical synthesis, crown ethers help stabilize reactive intermediates, making complex reaction mechanisms more efficient and allowing for the precise construction of medicinal compounds.
Electrolyte Systems in Batteries: Crown ethers play a role in enhancing the conductivity of electrolytes in lithium-ion and other advanced battery systems by coordinating with metal cations to improve ion transport.
Environmental Remediation: Crown ethers are employed in the extraction and sequestration of heavy metals from industrial wastewater, helping in environmental cleanup efforts by binding toxic ions and facilitating their removal.
Material Of The Product
Ethylene Oxide
A key starting material in the production of crown ethers, ethylene oxide undergoes polymerization to form the oxygen-containing rings that characterize crown ether structures.
Metal Ion Salts
Crown ethers are often used in combination with metal salts (such as sodium chloride or potassium iodide) to demonstrate their complexation capabilities in chemical synthesis and industrial applications.
Organic Solvents
Crown ethers are frequently dissolved in organic solvents such as dichloromethane, benzene, and ethanol, depending on their intended use in a reaction or process.
Benzene Rings (for Dibenzo Crown Ethers)
Benzene is used in the synthesis of dibenzo crown ethers, where the aromatic structure is integrated to enhance the compound's ability to bind specific ions.
Production Process Or Procedure
Crown ether production involves precise synthetic techniques to ensure the correct ring size and structure. Typically, crown ethers are synthesized through the cyclization of ethylene glycol derivatives with suitable electrophiles under controlled conditions. The process requires careful management of temperature and solvent systems to avoid side reactions that could lead to polymerization or incomplete ring closure. Additionally, high levels of purification are necessary to ensure the production of high-purity crown ethers suitable for industrial or pharmaceutical use.
Components Of The Product
Crown Ether Molecule
The central component of crown ether reactions, the cyclic polyether structure, selectively binds cations to facilitate various chemical processes, such as ion transport or catalysis.
Metal Cations (e.g., Na+, K+)
Metal cations are essential reactants in crown ether chemistry. Crown ethers specifically complex with these ions, affecting solubility and reactivity in many reactions.
Solvents
Organic solvents, such as acetone or dichloromethane, are commonly used to dissolve crown ethers and their reactants, enabling efficient reactions and separations.
Reaction By-products
During ion transport or separation processes, by-products may include uncomplexed ions or other reaction intermediates, which are often removed through filtration or crystallization.
Product Maintenance And Precautions
Proper Storage
Crown ethers should be stored in airtight containers, away from moisture, heat, and light, as these compounds are sensitive to environmental conditions that can affect their stability.
Handling Precautions
Wear protective gloves and safety goggles when handling crown ethers, as exposure to some crown ether compounds can cause skin and eye irritation.
Disposal
Waste crown ethers and any reaction by-products should be disposed of following local environmental regulations, particularly when dealing with metal-ion complexes.
Regular Inspection
Equipment used in crown ether reactions, such as reaction vessels and ion-separation setups, should be regularly inspected for signs of corrosion or buildup of by-products, ensuring proper functioning.
Company Advantages
Our Focus on Quality & Customer Experience
We dedicate to delivering high-quality products and exceptional customer service beyond your expectation.
Comprehensive Product Range
Extensive catalog of over 27,000 chemicals for diverse industries, offering both standard compounds and specialized solutions.
Strong Industry Expertise
Our experienced diverse team provides expert guidance, staying current with industry trends to deliver optimal chemical solutions tailored to your needs.
Competitive Pricing & Reliability
Competitive pricing and reliable deliveries ensure cost-effective, efficient supply chains without compromising quality.
How To Collaborate With Us
Partnering with LEAPChem gives you access to the expertise of a leading company with 18 years of experience in the chemical industry. We offer over 27,000 chemical substances tailored to meet diverse needs. All our products adhere to industry standards and are accompanied by the necessary certifications.
We collaborate with a wide range of partners, including:
• Research Laboratories
• Pharmaceutical Companies
• Cosmetics Manufacturers
• Electronics Manufacturers
• Chemical Manufacturers
• Universities and Colleges Worldwide
• And more.
We specialize in helping our clients find the right chemical solutions tailored to their specific requirements. Additionally, we provide custom synthesis services to meet the unique needs of our partners. Whether you need bulk quantities for industrial processes or small batches for specialized applications, we've got you covered.
If you are looking for a reliable chemical supplier, we would be delighted to hear from you. Please send your inquiry or project details to sales@leapchem.com. Our professional team is eager to collaborate with you and help achieve your goals.
FAQ
Q: What is Crown Ether?
A: Crown ether is a cyclic chemical compound that can bind metal ions, aiding in processes like ion transport, phase transfer catalysis, and chemical synthesis.
Q: What are crown ethers used for?
A: Crown ethers are used in ion complexation, organic synthesis, and as catalysts in phase transfer processes. They are also important in environmental remediation and battery technology.
Q: How does Crown Ether bind metal ions?
A: Crown ethers selectively bind metal ions based on their ring size, forming stable complexes by coordinating the metal ions within their cyclic structure.
Q: Why are crown ethers important in organic chemistry?
A: They enhance reaction rates and solubility of salts in organic solvents, facilitating reactions that would otherwise be difficult in non-aqueous systems.
Q: Which ions do crown ethers bind?
A: Crown ethers typically bind alkali metal ions like lithium, sodium, and potassium, with specific crown ethers designed to target different ions based on their size.
Q: Can crown ethers be reused?
A: Yes, in many cases, crown ethers can be recovered and reused in processes like ion separation, making them cost-effective for industrial applications.
Q: What is 18-Crown-6 used for?
A: 18-Crown-6 is particularly effective in binding potassium ions (K+) and is widely used in phase transfer catalysis and ion-selective applications.
Q: Are crown ethers safe to handle?
A: While generally stable, crown ethers can be irritants to skin and eyes. Proper protective equipment such as gloves and goggles should be worn when handling them.
Q: How are crown ethers made?
A: Crown ethers are synthesized by reacting ethylene glycol derivatives with electrophiles in the presence of solvents under controlled conditions.
Q: What role do crown ethers play in environmental cleanup?
A: Crown ethers are used to bind and remove toxic metal ions from wastewater, making them valuable for environmental remediation efforts.
Q: Are crown ethers used in batteries?
A: Yes, crown ethers improve electrolyte performance in batteries by enhancing the transport of metal ions, leading to better conductivity.
Q: How are crown ethers stored?
A: They are stored in airtight containers in cool, dry environments to prevent decomposition or reaction with moisture.
Q: What is phase transfer catalysis?
A: Phase transfer catalysis is a process where crown ethers help move ionic compounds from one phase (such as water) to another (such as an organic solvent), facilitating chemical reactions.
Q: What is the difference between crown ethers and azacrowns?
A: Azacrown ethers contain nitrogen atoms in their ring structure, allowing them to bind both cations and protons, unlike traditional crown ethers.
Q: Can crown ethers improve reaction efficiency?
A: Yes, crown ethers improve the efficiency of reactions by stabilizing intermediates and enhancing the solubility of ionic reactants in nonpolar solvents.
Hangzhou Leap Chem Co., Ltd. is well-known as one of the leading crown ether manufacturers and suppliers in China. We warmly welcome you to wholesale cheap crown ether from our factory. Contact us for custom service.
oled materials, 38304-91-5, cyclodextrin derivatives-
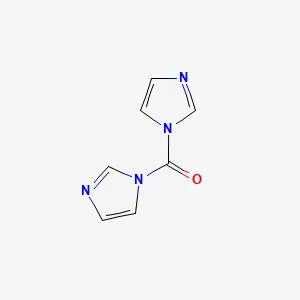
1,1'-Carbonyldiimidazole丨CAS 530-62-1Catalog No.: SS080884read more
CAS No.: 530-62-1
Assay(T): 98%min
Product Name:... -
Palladium丨CAS 7440-05-3Catalog No.: SS067788read more
CAS No.: 7440-05-3
Assay(Pd): 10% Pd/C on dry basis.
Product Name:... -
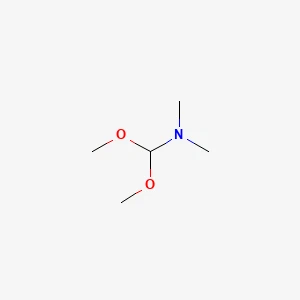
N,N-Dimethylformamide Dimethyl Acetal丨CAS 4637-24-5Catalog No.: SS036295read more
CAS No.: 4637-24-5
Assay: 98%min
Product Name: N,N-Dimethylformamide... -
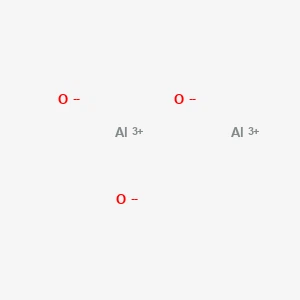
Aluminum Oxide丨CAS 1344-28-1Catalog No.: SS119815read more
CAS No.: 1344-28-1
Purity: 99.99% min; Average particle size: 50nm... -
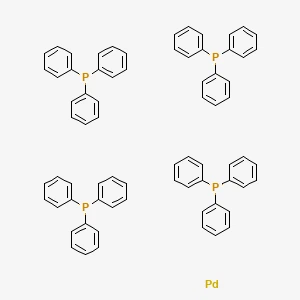
Tetrakis(triphenylphosphine)palladium丨CAS 14221-01-3Catalog No.: SS118525read more
CAS No.: 14221-01-3
Purity: 99%min Pd: 9.2%min
Product Name:... -
Alpha-Arbutin丨CAS 84380-01-8Catalog No.: SS087082read more
CAS No.: 84380-01-8
Purity: 99.5%min.
Product Name: alpha-Arbutin... -
N-Iodosuccinimide丨CAS 516-12-1Catalog No.: SS018455read more
CAS No.: 516-12-1
Purity: 98.0%min
Product Name: N-Iodosuccinimide... -
Octadecanethiol丨CAS 2885-00-9Catalog No.: SS085366read more
CAS No.: 2885-00-9
Assay(GC): 98.00%min
Product Name: Octadecanethiol...