Product Introduction
Ligands are molecules or ions that bind to a central metal atom or ion to form a coordination complex. In chemistry, ligands play a crucial role in stabilizing metal ions, facilitating chemical reactions, and modulating the properties of metal complexes. Ligands can be simple ions like chloride (Cl⁻) or complex organic molecules such as ethylenediamine (en) and porphyrins. Their ability to coordinate with metals makes them vital in catalysis, bioinorganic chemistry, material science, pharmaceuticals, and industrial chemical processes. Ligands come in various types, including monodentate, bidentate, and polydentate, based on the number of donor atoms they possess.
-
Isopropyltriphenylphosphonium Iodide丨CAS 24470-78-8Catalog No.: SS143173. CAS No.: 24470-78-8. Purity: 99.0% min/Assay: 98.0% min . Product Name: Isopropyltriphenylphosphonium iodide. Molecular Formula: C21H22P.I. Molecular Weight: 432.28. Synonym(s): (1-Methylethyl)triphenylphosphoniumread more
-
BI-Dime丨CAS 1373432-09-7Catalog No.: SS132672. CAS No.: 1373432-09-7. Purity(HPLC): 97%Min. Product Name: BI-Dime. Molecular Formula: C19H23O3P. Molecular Weight: 330.357881. Synonym(s): (S)-3-(tert-butyl)-4-(2,6-dimethoxyphenyl)-2,3-dihydrobenzod1,3oxaphosphole.read more
-
4,4'-Bi-1,3-benzodioxole-5,5'-diylbis(diphenylphosphine)丨...Catalog No.: SS132639. CAS No.: 210169-54-3. Purity(HPLC): 98%Min . Product Name: 4,4'-Bi-1,3-benzodioxole-5,5'-diylbis(diphenylphosphine). Molecular Formula: C38H28O4P2. Molecular Weight: 610.57. Synonym(s): (S)-SEGPHOS.read more
-
(R)-3-(tert-Butyl)-4-(2,6-dimethoxyphenyl)-2,2-dimethyl-2...Catalog No.: SS132471. CAS No.: 2634687-73-1. Purity: 97% Min. Product Name: (R)-3-(tert-Butyl)-4-(2,6-dimethoxyphenyl)-2,2-dimethyl-2,3-dihydrobenzo[d][1,3]oxaphosphole. Molecular Formula: C21H27O3P. Molecular Weight: 358.41 .read more
-
1,2-Bis((R)-phenyl(o-tolyl)phosphino)ethane丨CAS 81157-90-6Catalog No.: SS132407. CAS No.: 81157-90-6. Purity: 98%Min. Product Name: 1,2-Bis((R)-phenyl(o-tolyl)phosphino)ethane. Molecular Formula: C28H28P2. Molecular Weight: 426.5. Synonym(s): (R,R)-1,2-Bis(2-methylphenyl)(phenyl)phosphinoethane.read more
-
(R)-(6,6-Dimethoxy-1,1-biphenyl-2,2-diyl)bis(dicyclohexyl...Catalog No.: SS132395. CAS No.: 172617-14-0. Purity: 95%Min. Product Name: (R)-(6,6-Dimethoxy-1,1-biphenyl-2,2-diyl)bis(dicyclohexylphosphine) . Molecular Formula: C38H56O2P2. Molecular Weight: 606.8. Synonym(s):read more
-
(4S,5S)-1,3-Bis(2,2-diphenylethyl)-4,5-diphenyl-4,5-dihyd...Catalog No.: SS132328. CAS No.: 1033618-41-5. Purity: 97%Min. Product Name: (4S,5S)-1,3-Bis(2,2-diphenylethyl)-4,5-diphenyl-4,5-dihydro-1H-imidazol-3-ium tetrafluoroborate. Molecular Formula: C43H39BF4N2. Molecular Weight: 670.6.read more
-
7,9-Dimesityl-7H-acenaphtho1,2-dimidazol-9-ium Chloride丨C...Catalog No.: SS132293. CAS No.: 1286737-75-4 . Purity: 97%Min. Product Name: 7,9-Dimesityl-7H-acenaphtho1,2-dimidazol-9-ium chloride . Molecular Formula: C31H29ClN2. Molecular Weight: 465.03.read more
-
1,3-Dimesityl-4,5-dimethyl-1H-imidazol-3-ium Chloride丨CAS...Catalog No.: SS132290. CAS No.: 1118916-80-5. Purity(NMR): 95% Min. Product Name: 1,3-Dimesityl-4,5-dimethyl-1H-imidazol-3-ium chloride. Molecular Formula: C23H29ClN2. Molecular Weight: 368.95.read more
-
1,3-Bis(2,6-dibenzhydryl-4-methoxyphenyl)-1H-imidazol-3-i...Catalog No.: SS132289 . CAS No.: 1416368-03-0 . Purity(NMR): 97% Min. Product Name: 1,3-Bis(2,6-dibenzhydryl-4-methoxyphenyl)-1H-imidazol-3-ium chloride. Molecular Formula: C69H57ClN2O2. Molecular Weight: 981.68.read more
-
1,3-Bis(2,6-dibenzhydryl-4-methylphenyl)-1H-imidazol-3-iu...Catalog No.: SS132284. CAS No.: 1218778-19-8. Purity: 95%Min. Product Name: 1,3-Bis(2,6-dibenzhydryl-4-methylphenyl)-1H-imidazol-3-ium chloride. Molecular Formula: C69H57ClN2. Molecular Weight: 949.68. Synonym(s):read more
-
1-Mesityl-2,2,4-trimethyl-4-phenyl-3,4-dihydro-2H-pyrrol-...Catalog No.: SS132199. CAS No.: 1671098-42-2 . Purity: 95%Min . Product Name: 1-Mesityl-2,2,4-trimethyl-4-phenyl-3,4-dihydro-2H-pyrrol-1-ium tetrafluoroborate. Molecular Formula: C22H28BF4N . Molecular Weight: 393.28.read more
Product Advantages And Features
We are looking for a cooperative partner to expand our business.
Catalytic Efficiency
Ligands significantly enhance the efficiency of metal-catalyzed reactions by stabilizing transition states and facilitating selective transformations.
Selective Binding
Ligands can be designed to selectively bind specific metal ions, allowing for precise control over reaction outcomes in both industrial and laboratory settings.
Versatility in Coordination Chemistry
Ligands form complexes with various metal ions, enabling their use in a wide range of chemical reactions, including oxidation, reduction, and polymerization.
Modifiable Properties
By altering the ligand structure, chemists can fine-tune the reactivity, solubility, and stability of metal complexes, allowing for tailored applications in pharmaceuticals and catalysis.
Biological Relevance
Ligands are essential in biological systems, such as hemoglobin and chlorophyll, where they help transport oxygen and absorb light energy, respectively.
Improved Solubility and Stability
Ligands enhance the solubility and stability of metal ions in solution, making them easier to handle in industrial processes.
Product Type
Monodentate Ligands
These ligands contain a single donor atom that coordinates to a metal center. Examples include chloride (Cl⁻), ammonia (NH3), and water (H2O). They are widely used in basic coordination chemistry.
Bidentate Ligands
Bidentate ligands have two donor atoms, which allow them to form more stable chelate complexes with metal ions. Ethylenediamine (en) and oxalate (C2O4²⁻) are common examples.
Polydentate Ligands (Chelating Ligands)
These ligands have multiple donor atoms that can simultaneously bind to a metal center, creating a more stable complex. Examples include EDTA (ethylenediaminetetraacetate) and porphyrins.
Bridging Ligands
Bridging ligands coordinate to two or more metal atoms, linking them together in a complex. Examples include hydroxide (OH⁻) and carbonate (CO3²⁻).
Ambidentate Ligands
Ambidentate ligands have two potential binding sites, but they can only coordinate through one atom at a time. An example is the thiocyanate ion (SCN⁻), which can bind through sulfur or nitrogen.
Macrocyclic Ligands
These ligands contain large, ring-like structures that can encapsulate metal ions, offering exceptional stability. Crown ethers and porphyrins are examples of macrocyclic ligands.
Application Of The Product
Catalysis: Ligands are extensively used in catalysis, particularly in homogeneous catalysis, where they stabilize metal catalysts and influence reaction pathways. Examples include the use of phosphine ligands in palladium-catalyzed cross-coupling reactions.
Pharmaceuticals: Ligands are critical in the development of metal-based drugs, such as cisplatin for cancer treatment, where the ligand modulates the activity and selectivity of the drug.
Coordination Chemistry: Ligands are fundamental in forming coordination complexes, which are used in various chemical reactions, including synthesis and analysis.
Bioinorganic Chemistry: Ligands are essential in biological systems. For example, porphyrin ligands in hemoglobin coordinate with iron to enable oxygen transport in the bloodstream.
Material Science: Ligands are used in the production of advanced materials such as metal-organic frameworks (MOFs), which have applications in gas storage, separation, and catalysis.
Environmental Applications: Ligands are used to capture and remove metal ions from wastewater and to design sensors that detect environmental pollutants like heavy metals.
Electronics and Photonics: Ligands are used in the synthesis of metal complexes for organic light-emitting diodes (OLEDs) and photovoltaic cells, enhancing the performance of electronic devices.
Material Of The Product
Organic Molecules: Many ligands are derived from organic molecules, including phosphines, amines, and carboxylates. These ligands are synthesized for specific purposes, such as improving catalytic activity or selectivity.
Inorganic Ions: Simple inorganic ions, such as chloride (Cl⁻), cyanide (CN⁻), and sulfate (SO4²⁻), serve as ligands in coordination complexes and are widely used in both industrial and laboratory chemistry.
Macrocyclic Structures: Macrocyclic ligands, like crown ethers and porphyrins, are designed for high stability and specificity in binding metal ions, offering exceptional applications in catalysis and biological systems.
Phosphines: Phosphine ligands, such as triphenylphosphine (PPh3), are commonly used in metal-catalyzed reactions due to their electron-donating properties and ability to stabilize metal centers.
Amines and Amides: Ligands containing nitrogen donor atoms, such as ethylenediamine and pyridine, are highly effective at stabilizing transition metal complexes.
Carboxylates: Carboxylate ligands, such as acetate (CH3COO⁻), coordinate with metal ions through oxygen donor atoms and are essential in bioinorganic chemistry and catalysis.
Production Process Or Procedure
The synthesis of ligands involves precise organic or inorganic chemistry techniques, often tailored to the desired metal-binding properties and application. Ligands are designed to meet specific requirements, such as the number of donor atoms, flexibility, and electronic properties. Careful consideration of reaction conditions, including temperature, solvents, and purification methods, is essential to achieve high-purity ligands with consistent performance. Quality control through spectroscopic techniques like NMR, IR, and mass spectrometry ensures that ligands meet strict standards for industrial, pharmaceutical, and laboratory use.
Components Of The Product
Donor Atoms
Ligands coordinate to metal ions via donor atoms, such as nitrogen, oxygen, sulfur, or phosphorus, which provide lone pairs of electrons to form coordinate covalent bonds.
Organic Backbone
Many ligands have an organic framework, which can influence their flexibility, solubility, and binding properties. Examples include alkyl or aryl groups in phosphine ligands.
Functional Groups
Additional functional groups on ligands, such as hydroxyl (-OH) or carboxyl (-COOH) groups, enhance their reactivity and solubility in specific environments.
Chelating Moieties
Chelating ligands contain multiple donor atoms that can simultaneously bind to a metal ion, forming stable ring structures (chelate rings) that enhance the stability of the metal complex.
Macrocyclic Rings
Some ligands have large, cyclic structures (e.g., crown ethers, porphyrins), which form highly stable complexes with metal ions, often used in catalytic and biological applications.
Product Maintenance And Precautions
Ligands should be stored in airtight containers, away from moisture, light, and air, as some ligands, particularly organophosphines and thiol-based ligands, are sensitive to oxidation.
Ligands should be handled in inert environments (such as under nitrogen or argon), especially those that are sensitive to air or moisture, to maintain their reactivity.
Many ligand-metal complex reactions require strictly anhydrous conditions to prevent unwanted hydrolysis or oxidation of the ligands and metal centers.
Ligands should be periodically checked for purity using techniques such as NMR or HPLC, especially when used in high-precision applications like pharmaceuticals or catalysis.
Ensure that ligands are dissolved in compatible solvents that do not interfere with their activity or stability in chemical reactions.
Company Advantages
Our Focus on Quality & Customer Experience
We dedicate to delivering high-quality products and exceptional customer service beyond your expectation.
Comprehensive Product Range
Extensive catalog of over 27,000 chemicals for diverse industries, offering both standard compounds and specialized solutions.
Strong Industry Expertise
Our experienced diverse team provides expert guidance, staying current with industry trends to deliver optimal chemical solutions tailored to your needs.
Competitive Pricing & Reliability
Competitive pricing and reliable deliveries ensure cost-effective, efficient supply chains without compromising quality.
How To Collaborate With Us
Partnering with LEAPChem gives you access to the expertise of a leading company with 18 years of experience in the chemical industry. We offer over 27,000 chemical substances tailored to meet diverse needs. All our products adhere to industry standards and are accompanied by the necessary certifications.
We collaborate with a wide range of partners, including:
• Research Laboratories
• Pharmaceutical Companies
• Cosmetics Manufacturers
• Electronics Manufacturers
• Chemical Manufacturers
• Universities and Colleges Worldwide
• And more.
We specialize in helping our clients find the right chemical solutions tailored to their specific requirements. Additionally, we provide custom synthesis services to meet the unique needs of our partners. Whether you need bulk quantities for industrial processes or small batches for specialized applications, we've got you covered.
If you are looking for a reliable chemical supplier, we would be delighted to hear from you. Please send your inquiry or project details to sales@leapchem.com. Our professional team is eager to collaborate with you and help achieve your goals.
FAQ
Q: What are ligands in chemistry?
A: Ligands are molecules or ions that bind to a central metal atom to form coordination complexes, essential in catalysis, pharmaceuticals, and bioinorganic chemistry.
Q: How do ligands bind to metals?
A: Ligands bind to metals through donor atoms, which provide lone pairs of electrons to form coordinate covalent bonds with the metal center.
Q: What is a monodentate ligand?
A: A monodentate ligand contains one donor atom that coordinates to a single metal atom, forming a simple coordination bond.
Q: What is a chelating ligand?
A: A chelating ligand has multiple donor atoms that can form more than one bond to a metal ion, creating stable ring-like structures known as chelates.
Q: What are some common ligands used in catalysis?
A: Common ligands in catalysis include phosphines (e.g., PPh3), amines (e.g., ethylenediamine), and macrocyclic ligands (e.g., crown ethers).
Q: What is the importance of ligands in biology?
A: In biological systems, ligands, such as porphyrins, play a critical role in functions like oxygen transport (hemoglobin) and photosynthesis (chlorophyll).
Q: Can ligands be modified for specific applications?
A: Yes, ligands can be chemically tailored to modify their binding properties, selectivity, and reactivity for specific industrial, pharmaceutical, or catalytic applications.
Q: What are ambidentate ligands?
A: Ambidentate ligands have two potential binding sites but can only bind through one donor atom at a time, such as the thiocyanate ion (SCN⁻).
Q: Why are macrocyclic ligands more stable?
A: Macrocyclic ligands create more stable metal complexes due to their ring-like structure, which encapsulates the metal ion, providing enhanced stability.
Q: What role do ligands play in pharmaceutical chemistry?
A: Ligands are used to develop metal-based drugs and influence their activity, targeting specific biological processes or pathogens.
Q: What is the difference between polydentate and bidentate ligands?
A: Bidentate ligands have two donor atoms that bind to a metal, while polydentate ligands have more than two, forming even more stable complexes.
Q: What safety precautions should be taken when handling ligands?
A: Handle ligands under inert conditions, store them properly, and use appropriate solvents and protective equipment to prevent degradation or unwanted reactions.
Q: Can ligands be used in environmental applications?
A: Yes, ligands are used in environmental chemistry to remove heavy metals from wastewater and in designing sensors to detect pollutants.
Q: What is the role of phosphine ligands in catalysis?
A: Phosphine ligands are widely used in transition metal catalysis due to their ability to donate electrons and stabilize metal catalysts in reactions like cross-coupling.
Q: What are the byproducts of ligand-metal reactions?
A: Byproducts of ligand-metal reactions vary but often include salts, unreacted ligands, or side products formed during the formation of coordination complexes.
Hangzhou Leap Chem Co., Ltd. is well-known as one of the leading ligands manufacturers and suppliers in China. We warmly welcome you to wholesale cheap ligands from our factory. Contact us for custom service.
41621-49-2, CAS 26016 99 9, antibiotics-
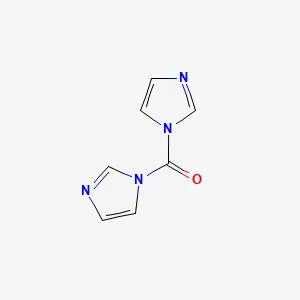
1,1'-Carbonyldiimidazole丨CAS 530-62-1Catalog No.: SS080884read more
CAS No.: 530-62-1
Assay(T): 98%min
Product Name:... -
Palladium丨CAS 7440-05-3Catalog No.: SS067788read more
CAS No.: 7440-05-3
Assay(Pd): 10% Pd/C on dry basis.
Product Name:... -
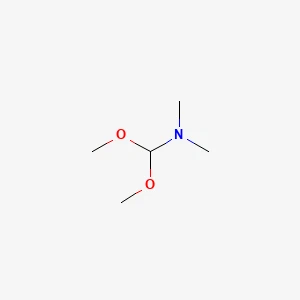
N,N-Dimethylformamide Dimethyl Acetal丨CAS 4637-24-5Catalog No.: SS036295read more
CAS No.: 4637-24-5
Assay: 98%min
Product Name: N,N-Dimethylformamide... -
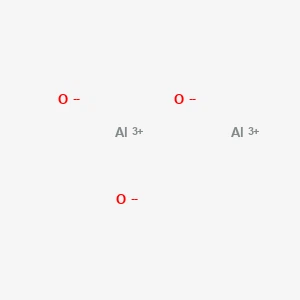
Aluminum Oxide丨CAS 1344-28-1Catalog No.: SS119815read more
CAS No.: 1344-28-1
Purity: 99.99% min; Average particle size: 50nm... -
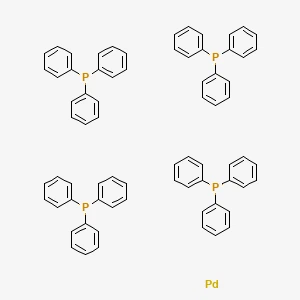
Tetrakis(triphenylphosphine)palladium丨CAS 14221-01-3Catalog No.: SS118525read more
CAS No.: 14221-01-3
Purity: 99%min Pd: 9.2%min
Product Name:... -
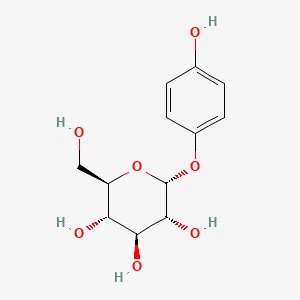
Alpha-Arbutin丨CAS 84380-01-8Catalog No.: SS087082read more
CAS No.: 84380-01-8
Purity: 99.5%min.
Product Name: alpha-Arbutin... -
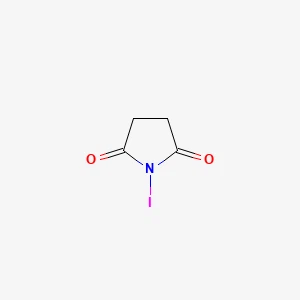
N-Iodosuccinimide丨CAS 516-12-1Catalog No.: SS018455read more
CAS No.: 516-12-1
Purity: 98.0%min
Product Name: N-Iodosuccinimide... -
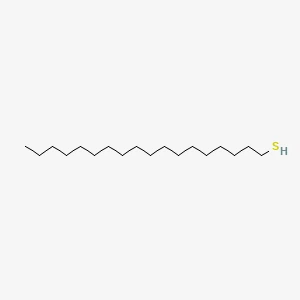
Octadecanethiol丨CAS 2885-00-9Catalog No.: SS085366read more
CAS No.: 2885-00-9
Assay(GC): 98.00%min
Product Name: Octadecanethiol...




![(R)-3-(tert-Butyl)-4-(2,6-dimethoxyphenyl)-2,2-dimethyl-2,3-dihydrobenzo[d][1,3]oxaphosphole丨CAS 2634687-73-1](/uploads/42644/small/r-3-tert-butyl-4-2-6-dimethoxyphenyl-2-2a9b00.png?size=380x0)