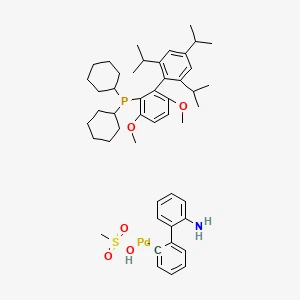
Hangzhou Leap Chem Co., Ltd. is one of the most professional manufacturers and suppliers of brettphos pd g3丨cas 1470372-59-8 in China. Welcome to wholesale custom made chemical products at competitive price from our factory. For more cheap products, contact us now.
Specifications of BrettPhos Pd G3丨CAS 1470372-59-8
|
Appearance |
Yellow to brown powder |
|
Structural Identification |
HNMR |
|
Purity |
98% min |
|
Insolubles (in DCM) |
1% max |
|
Storage Condition |
Keep container tightly closed, avoid direct sunlight. Avoid contact with oxidizing agents and high temperature. |
Transport Information of BrettPhos Pd G3丨CAS 1470372-59-8
|
Parameter |
Specification |
|
UN Number |
|
|
Class |
|
|
Packing Group |
|
|
H.S. Code |
3815120090302 |
|
Stability & Reactivity |
The product is chemically stable under standard ambient conditions. |
|
Storage |
2~8 deg.C |
|
Condition to Avoid |
|
|
Package |
Overview
BrettPhos Pd G3丨CAS 1470372-59-8 is a third-generation, preformed palladium precatalyst complexed with the BrettPhos ligand. Developed for cross-coupling reactions, it allows for high-efficiency C–N, C–O, and C–C bond formation under mild conditions. It is air and moisture stable, easy to handle, and widely used in academic research and pharmaceutical process development.
Applications
1. Buchwald–Hartwig Amination
Efficient for forming C–N bonds between (hetero)aryl halides and amines, including:
Primary and secondary alkyl amines
Aromatic amines
Hindered or electron-deficient substrates
2. Aryl Ether Formation (C–O Coupling)
Facilitates coupling of phenols or alcohols with aryl halides, useful in synthesis of:
Pharmaceuticals
Agrochemicals
Natural products
3. Suzuki–Miyaura Coupling
Supports aryl–aryl cross-coupling using boronic acids, although its use here is less common compared to its C–N/C–O scope.
4. Complex Molecule Construction
Preferred in the synthesis of medicinal compounds, bioactive scaffolds, and functionalized aromatic structures requiring:
High selectivity
Functional group tolerance
Mild reaction conditions
Benefits
✅ Air- and Moisture-Stable Solid
Can be handled on the bench without a glovebox or inert atmosphere, enabling easier lab-scale and process use.
✅ High Catalytic Efficiency
Active at low catalyst loadings (as low as 0.1–1 mol%), even with sterically hindered or deactivated substrates.
✅ Excellent Substrate Scope
Performs well with electron-poor, electron-rich, and sterically hindered aryl halides - compatible with a wide variety of nucleophiles.
✅ Reproducibility and Scalability
Used widely in industrial and pharmaceutical synthesis due to its robust and predictable behavior.
✅ Mild Conditions
Reactions can proceed at moderate temperatures, minimizing decomposition of sensitive reactants or products.
Summary
BrettPhos Pd G3丨CAS 1470372-59-8 is a third-generation palladium pre-catalyst designed for efficient cross-coupling, especially C–N and C–O bond formation. Its unique combination of stability, low loading requirements, broad substrate tolerance, and user-friendliness makes it a preferred tool in modern synthetic and medicinal chemistry.